
Denaturation of protein is a common occurrence during food preparation and cooking. Proteins are large, complex molecules composed of chains of amino acids folded into distinct 3D shapes. In this state, they are called native folded protein, and are able to perform their specific biological functions. But when they are denatured, they lose their functions. Denaturation is a process that disrupts the structure of a protein, causing it to lose shape and function.
Denaturation in the context of food is frequently brought on by heat, acidity, or mechanical agitation. As an illustration, when an egg is cooked, the heat causes the egg white’s proteins to denature and coagulate, resulting in the formation of a solid mass. This is similar to how milk denatures and coagulates when exposed to acid, such as vinegar or lemon juice, to form curds.
Denaturation can affect the value of foods in both good and bad ways. Proteins in meat, for instance, become denatured during cooking in order to make them more digestible. Protein breakdown can also result in the release of new flavors and aromas. The extent of these changes is determined by the type of protein in the food as well as the degree of denaturation.
But excessive denaturation can also lead to nutrient loss and changes in texture and flavor. In some instances, the food can no longer be accepted as an ingredient. When frozen egg yolks are thawed, they become lumpy and unpalatable because their lipoprotein has denatured and aggregated.In protein drinks, high protein solubility and dispersibility are necessary. Hence, partial protein denaturation during processing may cause flocculation and precipitation during storage, affecting the product’s sensory attributes.
Understanding the science behind protein denaturation is therefore critical for optimizing food quality and nutritional value.
Let’s discuss further.
WHAT IS PROTEIN DENATURATION?
The intricacy of many food systems makes it unsurprising that denaturation is a complex process that is not easily explained in simple terms. Denaturation of proteins involves the alteration of their structure, causing them to lose their shape and function. This can occur when proteins are subjected to heat, acid, or other environmental factors. Denaturation of a protein leads to its inability to perform its original function, and this can result in changes to the texture and flavor of the food it is in. For example, enzymes, which are a type of protein, lose their catalytic ability and become inactive when they are denatured.
Here is what exactly happens during denaturation of protein.
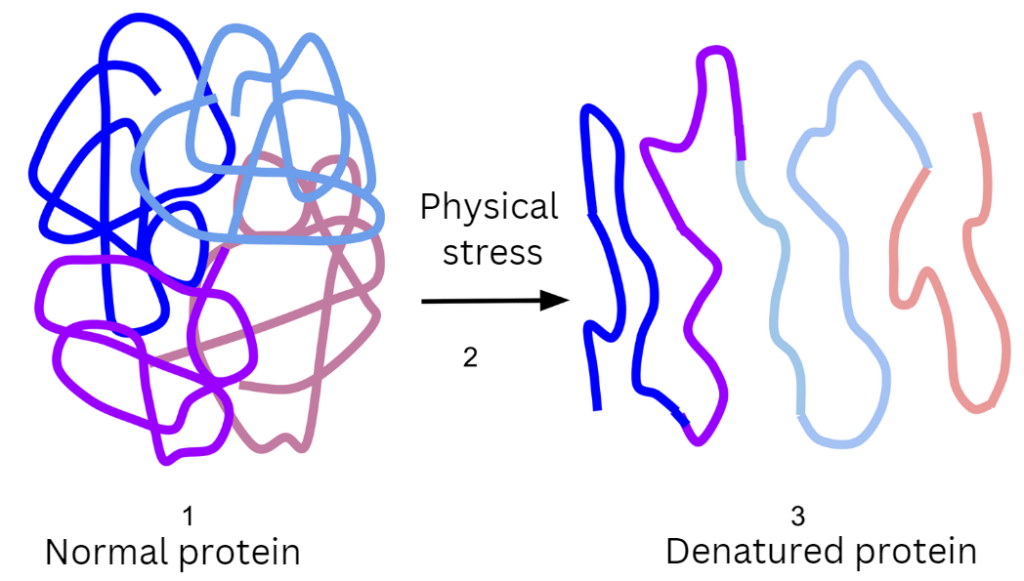
When a protein becomes denatured, there is a change in its secondary, tertiary, and/or quaternary structure. Its primary structure is not altered. Hence, there is no breaking of the peptide bonds. Peptide bonds are chemical covalent bonds that connect amino acids in a protein chain. Peptide bonds are extremely strong and stable, and they play an important role in protein stability and function. A protein chain’s backbone is made up of a repeating sequence of peptide bonds, which give the protein its distinctive shape and stability. When the protein is denatured, it does unfold, but the amino acid sequence remains the same.
When hydrogen bonds, ionic bonds, or hydrophobic interactions within a protein are broken by a treatment, denaturation occurs. Denaturation can often be reversed if the denaturing agent stabilizes the protein in its unfolded state, allowing the protein to refold back to its native conformation when the agent is removed. However, denaturation can also be irreversible if an unfolded protein is stabilized by interaction with other protein chains, such as with egg proteins during boiling.
FACTORS THAT AFFECT PROTEIN DENATURATION IN FOOD
Various factors can cause protein denaturation. Some of these factors include: heat, freezing, pH change, ionic strength change, and surface changes.
Heat
Denaturation is typically the result of mild changes. Hence, mild heat treatments such as pasteurization or blanching can induce protein denaturation..
Meat proteins are denatured at temperatures ranging from 135°F (57°C) to 167°F (75°C), which has a significant impact on the texture, water holding capacity, and shrinkage. The native proteins in meat keep the tissue moist after cutting or grinding. When heated, the proteins denature and have fewer interactions with water. The juices that leak from a cooked steak are mostly water that is no longer retained by myosin proteins. Resting a steak after cooking allows some of the water to find new interactions with proteins, resulting in less juice leaking out when the steak is cut.
Heat usually causes a change in the tertiary structure, resulting in a less ordered arrangement of the polypeptide chains. But there are exceptions to this.
Milk casein and gelatin are proteins that are quite heat stable. And they can be heat treated without apparent change in their stability. Casein’s exceptional stability makes it possible to boil, sterilize, and concentrate milk without causing coagulation. The restrictions against the formation of a folded tertiary structure explain the heat stability of these proteins. The relatively high proline and hydroxyproline content in the heat stable proteins is what causes these restrictions.
The likelihood of inter- and intramolecular hydrogen bonds forming is higher in peptide chains lacking proline than in chains with numerous proline residues. This demonstrates how the secondary and tertiary protein structures, which in turn are in charge of the protein’s functionality, are directly related to the amino acid composition of proteins.
Freezing
Protein denaturation can occur not only during thermal processing of foods, it also occurs at low temperatures. Exposing food to freezing temperatures ranging from 32°F (0°C) to 5°F (-15°C) also induces denaturation of proteins. Muscle proteins are more susceptible to denaturation than plant-derived proteins. At low temperatures, ice crystals form, which can rupture cell membranes, followed by denaturation due to surface phenomena at the interface of the two phases (protein solution and ice crystals), or possibly due to the freezing of water required to maintain the protein’s native conformation.
One way to minimize denaturation is freezing the food at a fast freezing rate, which is usually achieved by industrial freezers.
You might also like: Why You Should Not Freeze Meat At Home
In this manner, the interior moisture of the cell is rapidly cooled, resulting in the formation of small ice crystals, usually spearlike and separated by proteins, inside the cell. As a result, there is less dehydration and damage to proteins. Slow freezing, in contrast, often results in dehydration and an increase in the cell’s ionic strength. Water drawn from the cell’s interior will freeze onto the already existing extracellular ice crystals, causing them to grow and distort and damage the membrane and proteins.
In the fish industry, cryoprotectants such as polyols or antioxidants are applied prior to freezing. Doing so helps preserve protein stability and functionality during freezing and storage.
pH change
pH is an important element that can influence protein denaturation in food. The pH of a food product can influence the charge of the amino acid residues within a protein molecule, which affects the shape, stability, and function of the protein. When the pH of a food changes, the protein molecules might change shape, resulting in protein denaturation.
When proteins in food are exposed to acidic conditions, such as those present in citrus fruits or vinegar, they may denature. Because of the acidic environment, the protein molecules unfold, losing their functional structure and rendering them incapable of performing their intended function. However, in some cases, such as when making cheese, this is beneficial.

In cheese manufacturing, milk is often acidified by using lactic acid bacteria, which convert lactose (a sugar in milk) to lactic acid. The pH of milk decreases as the lactic acid concentration increases, causing the casein proteins to become less soluble and coagulate. The coagulation process separates the solid curds from the liquid whey, which is necessary for the creation of cheese.
This acid-induced protein denaturation also adds to the distinct texture and flavor of various cheeses. Fresh cheese, for example, is prepared by boiling milk and acidifying it with vinegar or citric acid. Because of the acidity, the proteins denature and produce small, fragile curds that are separated from the liquid whey. Hard, aged cheese, on the other hand, goes through a longer acidification process, resulting in more extensive protein denaturation and a firmer texture and sharper flavor.
Ionic strength change
Ionic strength refers to the concentration of ions in a solution, which can be increased by adding salt. The ionic strength of a solution affects protein denaturation by altering the strength of electrostatic interactions between protein molecules. Generally, increasing the ionic strength of a food can reduce the extent of protein denaturation. This is because high ionic strength solutions weaken electrostatic connections between protein molecules, thereby reducing their ability to denature and aggregate.
Salts can be utilized to induce protein denaturation and gel formation in various food processing applications, such as meat curing. Salts increase the ionic strength of the solution in certain circumstances, resulting in more extensive protein denaturation and aggregation. This can result in the production of a gel-like structure, which is desired in some processed meat products.
Surface changes
Surface change can influence protein denaturation by changing the interactions between proteins and their surroundings. Proteins have several interfaces that allow them to interact with water molecules, other proteins, and other molecules in their surroundings. These surface interactions can affect the protein’s stability and structure.
One example of surface change is brought about by altering the hydrophobicity (ability of a substance to repel water) of the protein surface. Proteins have hydrophobic and hydrophilic surfaces that influence how they interact with water molecules and other proteins. The presence of specific chemicals or changes in the polarity of the environment might modify the hydrophobicity of the protein surface, reducing its stability and leading to denaturation.
Changing the polarity of the protein can also influence protein denaturation. The presence of particular solvents or changes in pH can alter the charge distribution on the protein surface, influencing the strength and specificity of electrostatic interactions between proteins and other molecules.
You might also like: How Whipping Turns Egg Whites Into A Foam
The most commonly observed surface change in food is the process of beating egg whites. Egg whites contain a globular, water-soluble protein called albumin. When eggs are beaten, the albumin molecules unfold, exposing their hydrophobic amino acid residues to the air. This allows the albumin molecules to incorporate air into the mixture. As a result, the protein becomes more hydrophobic, creating a new surface area that can interact with other hydrophobic amino acids on nearby protein molecules. These protein networks stabilize the air bubbles and prevent them from escaping, resulting in a stable foam structure. This foam structure is useful for making cakes, meringues, and soufflés due to its high volume and light texture.
APPLICATIONS OF PROTEIN DENATURATION IN FOOD
Protein denaturation is better understood from a food manufacturing perspective, as the food industry relies on manipulating proteins to produce desirable food products. However, it is also important during cooking and food preparation, as it occurs almost every time. Denaturing protein during cooking makes meat more palatable and easier to digest. In food manufacturing, protein denaturation occurs during various processes. Here are a few examples:
One of the most common applications of protein denaturation is cheese manufacturing. Adding an acid during the cheese-making process denatures the proteins in milk, resulting in the formation of curds. After being separated from the liquid whey, the curds are utilized to manufacture a variety of cheese products. Milk protein denaturation is critical for cheese formation and is tightly monitored to ensure consistent quality and product characteristics.

Protein denaturation is also essential in the creation of foams like whipped cream or meringue. Proteins must denature and form a network capable of stabilizing air bubbles in order to build a stable foam structure. Controlling the quantity of protein denaturation during foam generation is crucial for getting the optimum foam texture and stability. Mixing time, temperature, and pH can all influence the extent of protein denaturation and, as a result, the end product’s quality.
Protein denaturation is also used in the manufacturing of emulsions such as mayonnaise and salad dressings. Emulsions are often created by blending oil and water, which do not mix naturally. Denatured proteins, on the other hand, can stabilize the emulsion by enveloping and stabilizing oil droplets in a water-based solution. The emulsion’s stability is affected by a number of parameters, including the type and concentration of proteins used, as well as pH and temperature.
References:
M. Gibson (2018). Food Science and the Culinary Arts. Academic Press.
V. Vaclavik, E. Christian (2014). Essentials of Food Science (4th edition). Springer.
P. Cheung, B. Mehta (2015). Handbook of Food Chemistry. Springer
J. Provost, K. Colabroy, B. Kelly, M. Wallert (2016). The Science of Cooking: Understanding the Biology and Chemistry Behind Food and Cooking. John Wiley & Sons, Inc.
J. deMan, J. Finley, W. Jeffrey Hurst, C. Y. Lee (2018). Principles of Food Chemistry (4th edition). Springer.
H. Belitz, W. Grosch, P. Schieberle (2009). Food Chemistry (4th Edition). Springer


